Silver Atom
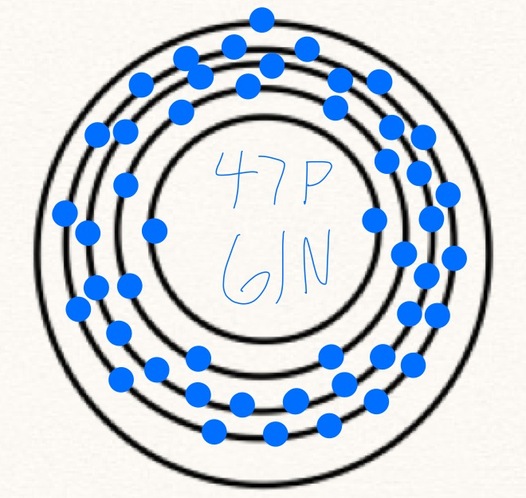
This is the atom of silver. If you have not read the basic information page, then you might want to look at it because it will help with this. From that page we concluded that there are 47 electrons, 47 protons and 61 neutrons. When drawing the silver atom, you simply start with 5 rings because silver is in the 5th period on the periodic table. Since you have 47 electrons, you put 47 dots on the rings, but you can only put 2 on the first ring, 8 on the second, 18 on the third and 18 on the fourth. If you add that up, you get 46 so you have one more electron which you can just put on the last ring. In the middle (the nucleus) you write 47 P for the 47 protons and 61 N for the 61 neutrons. If you do that, you will have a silver atom model. Please note that this isn't actually what a silver atom looks like, this is just a model that helps us understand the components of the atom. Please also note that this isn't the only isotope of silver, this is just the most common.
